Alzheimer’s Disease-Related Copper(II)-β-Amyloid
Peptide Exhibits Phenol Monooxygenase and Catechol Oxidase Activities**
Giordano F. Z. da Silva and Li-June Ming
Angew. Chem. Int. Ed. 2005, 44, 5501–5504. (Accepted without revision.)
The neuropathology of Alzheimer’s disease (AD) is attributed to the 40 amino
acid amyloid-β peptide which binds redox-active metal ions, such as in CuII,
with concomitant aggregation and generation of reactive oxygen species (ROS).
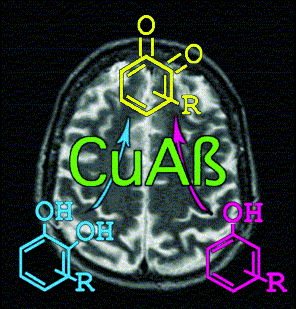
We have observed that the CuII complex of a C-terminal
truncated amyloid-β (CuAβ1-20) is soluble and able
to catalyze reactions consistent with the activities of the Type-3 copper
enzymes phenol monooxygenase and catechol oxidase. The oxidation of
catechol to o-quinone catalyzed by CuAβ1-20
shows remarkable rate accelerations of 3.25 × 105
and 1.12 × 106 in terms of the first-order
rate constant kcat in aqueous solution at pH 7.0 in the absence and presence
of 25 mM H2O2, respectively.
More significantly, the relatively more inert phenol can be hydroxylated and
oxidized by CuAβ1-20 to yield o-quinone with
8.67 × 104 and 4.60 × 106
rate accelerations and deuterium kinetic isotope effect of 3.46 and 4.77 in
the absence and presence of 50 mM H2O2,
respectively, at pH 7.0,. These data expose novel Cu-oxygen chemistry
of CuAβ1-20 consistent with Type-3 copper enzymes
which offer better understanding of the chemical effect in the brains of AD
patients and possible treatment strategies of this disease.
(reprint
in pdf)
**The authors dedicate this work to the Kelley
and Sotolongo families for their unfortunate experience with Alzheimer’s
disease. Our research on the structure and activity of metallopeptides
was partially supported by the Research and Creative Award of the University
of South Florida and the Petroleum Research Fund administrated by the American
Chemical Society (PRF AC #40851-AC3).We thank John Kelley for the modification
of the picture for the Table of Contents.